Amino acid and neurotransmitter
- For the anion in its role as a neurotransmitter, see Glutamate (neurotransmitter).
IUPAC name Glutamic acid
Systematic IUPAC name 2-Aminopentanedioic acid
Other names
- 2-Aminoglutaric acid
CAS Number
- l isomer: 56-86-0
- racemate: 617-65-2
- d isomer: 6893-26-1
3D model (JSmol)
- l isomer: Interactive image
- d isomer: Interactive image
- Zwitterion: Interactive image
- Deprotonated zwitterion: Interactive image
Beilstein Reference
1723801 (L) 1723799 (rac) 1723800 (D)
ChEBI
- l isomer: CHEBI:16015
- racemate: CHEBI:18237
- d isomer: CHEBI:15966
ChEMBL
- l isomer: ChEMBL575060
ChemSpider
- l isomer: 30572
DrugBank
- l isomer: DB00142
- d isomer: DB02517
ECHA InfoCard
100.009.567
EC Number
- l isomer: 200-293-7
E number
E620 (flavour enhancer)
Gmelin Reference
3502 (L) 101971 (rac) 201189 (D)
KEGG
- l isomer: C00025
- d isomer: C00217
PubChem CID
- l isomer: 33032
- d isomer: 23327
UNII
- l isomer: 3KX376GY7L
- racemate: 61LJO5I15S
- d isomer: Q479989WEA
CompTox Dashboard (EPA)
- l isomer: DTXSID0046987
InChI
InChI=1S/C5H9NO4/c6-3(5(9)10)1-2-4(7)8/h3H,1-2,6H2,(H,7,8)(H,9,10)Key:WHUUTDBJXJRKMK-UHFFFAOYSA-N
- l isomer: InChI=1/C5H9NO4/c6-3(5(9)10)1-2-4(7)8/h3H,1-2,6H2,(H,7,8)(H,9,10)Key:WHUUTDBJXJRKMK-UHFFFAOYAD
- l isomer: C(CC(=O)O)C@@HN
- d isomer: C(CC(=O)O)C@HN
Zwitterion: C(CC(=O)O)C(C(=O)[O-])[NH3+]
Deprotonated zwitterion: C(CC(=O)[O-])C(C(=O)[O-])[NH3+]
Chemical formula
C5H9NO4
Molar mass
147.130g·mol
Appearance
White crystalline powder
Density
1.4601 (20°C)
Melting point
199°C (390°F; 472K) decomposes
Solubility in water
8.57g/L (25 °C)
Solubility
Ethanol: 350μg/100g (25°C)
Acidity (pKa)
- 2.10 (α-carboxyl; H2O)
- 4.07 (side chain; H2O)
- 9.47 (α-amino; H2O)
Magnetic susceptibility ()
−78.5·10 cm/mol
Hazards
GHS labelling:
Pictograms
Signal word
Warning
Hazard statements
H315, H319, H335
Precautionary statements
P261, P264, P271, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501
NFPA 704 (firediamond)
2 1 0
Supplementary data page
Glutamic acid (data page)
Except where otherwise noted, data are given for materials in their standard state (at 25°C [77°F], 100kPa).
Infobox references
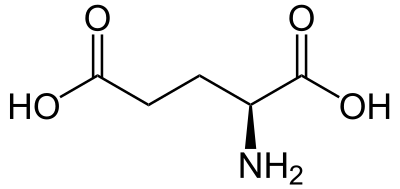
Glutamic acid (symbol Glu or E; known as glutamate in its anionic form) is an α-amino acid that is used by almost all living beings in the biosynthesis of proteins. It is a non-essential nutrient for humans, meaning that the human body can synthesize enough for its use. It is also the most abundant excitatory neurotransmitter in the vertebrate nervous system. It serves as the precursor for the synthesis of the inhibitory gamma-aminobutyric acid (GABA) in GABAergic neurons.
Its molecular formula is C 5H 9NO 4. Glutamic acid exists in two optically isomeric forms; the dextrorotary L-form is usually obtained by hydrolysis of gluten or from the waste waters of beet-sugar manufacture or by fermentation. Its molecular structure could be idealized as HOOC−CH(NH 2)−(CH 2)2−COOH, with two carboxyl groups −COOH and one amino group −NH 2. However, in the solid state and mildly acidic water solutions, the molecule assumes an electrically neutral zwitterion structure OOC−CH(NH 3)−(CH 2)2−COOH. It is encoded by the codons GAA or GAG.
The acid can lose one proton from its second carboxyl group to form the conjugate base, the singly-negative anion glutamate OOC−CH(NH 3)−(CH 2)2−COO. This form of the compound is prevalent in neutral solutions. The glutamate neurotransmitter plays the principal role in neural activation. This anion creates the savory umami flavor of foods and is found in glutamate flavorings such as monosodium glutamate (MSG). In Europe, it is classified as food additive E620. In highly alkaline solutions the doubly negative anion OOC−CH(NH 2)−(CH 2)2−COO prevails. The radical corresponding to glutamate is called glutamyl.
The one-letter symbol E for glutamate was assigned as the letter following D for aspartate, as glutamate is larger by one methylene –CH2– group.